| Species Reactivity | Human |
| Clone | petosemtamab |
| Source/Isotype | Human IgG1(K214R,L351D,D356E,L358M,L368E,delta447K), Kappa |
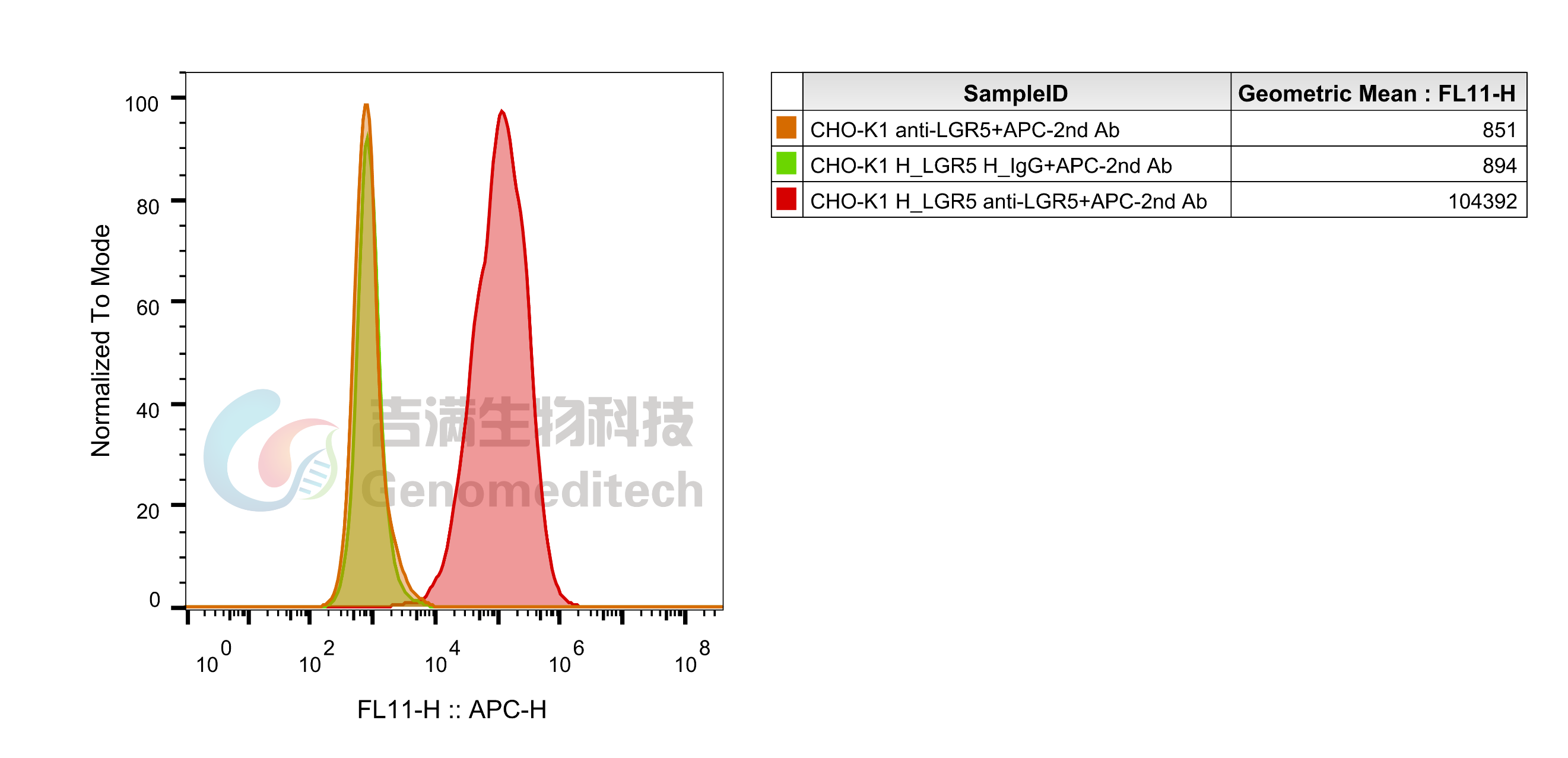
| Application | Bioactivity-ELISA; Flow Cytometry |
| Target | Detects EGFR&LGR5 |
| Gene | EGFR&LGR5 |
| Other Names | EGFR:ERBB, ERBB1, ERRP, HER1, NISBD2, NNCIS, PIG61, mENA LGR5:FEX, GPR49, GPR67, GRP49, HG38 |
| Gene ID | EGFR: 1956 (Human) LGR5: 8549 (Human) |
| Background | Petosemtamab (MCLA-158) is an EGFR/LGR5 bispecific monoclonal antibody drug with a research background derived from the unmet need that exists in the treatment of solid tumors such as head and neck squamous-cell carcinoma (HNSCC) . EGFR, as a key driver of tumor growth, is overexpressed in HNSCC, while LGR5, as a cancer stem cell marker, is closely related to tumor recurrence and metastasis. Traditional single-targeted drugs can not simultaneously block EGFR signaling and eliminate LGR5 + cancer stem cells, resulting in limited efficacy and drug resistance. PETOSEMTAMAB, developed through the Biclonics dual antibody platform, has a triple mechanism of action: direct blockade of the EGFR signaling pathway, induction of LGR5-mediated endocytic degradation of EGFR, and enhancement of antibody-dependent cytotoxicity (ADCC) and phagocytosis (ADCP) , which may be beneficial for the development of anti-EGFR drugs, thus, multi-dimensional inhibition of tumor growth and metastasis is achieved. Preclinical studies have shown that the drug significantly inhibits tumor growth and does not interfere with the function of healthy stem cells in models of head and neck cancer and colorectal cancer. |
| Storage | Store at 2-8℃ short term (1-2 weeks).Store at ≤ -20℃ long term. Avoid repeated freeze-thaw. |
| Formulation | Phosphate-buffered solution, pH 7.2-7.4. |
| Endotoxin | < 1 EU/mg, determined by LAL gel clotting assay |
| Species Reactivity | Human |
| Clone | petosemtamab |
| Source/Isotype | Human IgG1(K214R,L351D,D356E,L358M,L368E,delta447K), Kappa |
| Application | Bioactivity-ELISA; Flow Cytometry |
| Target | Detects EGFR&LGR5 |
| Gene | EGFR&LGR5 |
| Other Names | EGFR:ERBB, ERBB1, ERRP, HER1, NISBD2, NNCIS, PIG61, mENA LGR5:FEX, GPR49, GPR67, GRP49, HG38 |
| Gene ID | EGFR: 1956 (Human) LGR5: 8549 (Human) |
| Background | Petosemtamab (MCLA-158) is an EGFR/LGR5 bispecific monoclonal antibody drug with a research background derived from the unmet need that exists in the treatment of solid tumors such as head and neck squamous-cell carcinoma (HNSCC) . EGFR, as a key driver of tumor growth, is overexpressed in HNSCC, while LGR5, as a cancer stem cell marker, is closely related to tumor recurrence and metastasis. Traditional single-targeted drugs can not simultaneously block EGFR signaling and eliminate LGR5 + cancer stem cells, resulting in limited efficacy and drug resistance. PETOSEMTAMAB, developed through the Biclonics dual antibody platform, has a triple mechanism of action: direct blockade of the EGFR signaling pathway, induction of LGR5-mediated endocytic degradation of EGFR, and enhancement of antibody-dependent cytotoxicity (ADCC) and phagocytosis (ADCP) , which may be beneficial for the development of anti-EGFR drugs, thus, multi-dimensional inhibition of tumor growth and metastasis is achieved. Preclinical studies have shown that the drug significantly inhibits tumor growth and does not interfere with the function of healthy stem cells in models of head and neck cancer and colorectal cancer. |
| Storage | Store at 2-8℃ short term (1-2 weeks).Store at ≤ -20℃ long term. Avoid repeated freeze-thaw. |
| Formulation | Phosphate-buffered solution, pH 7.2-7.4. |
| Endotoxin | < 1 EU/mg, determined by LAL gel clotting assay |