| Species Reactivity | Human |
| Clone | Vilobelimab |
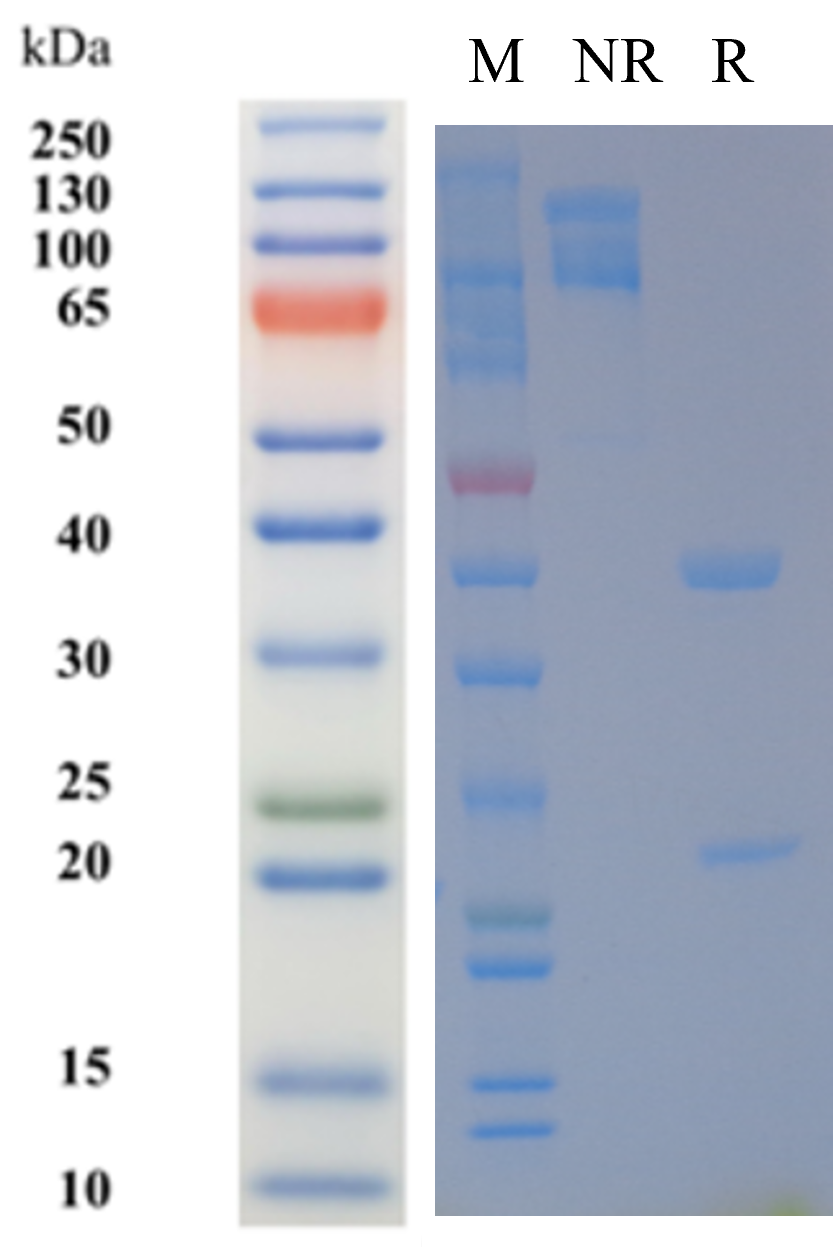
| Source/Isotype | Human IgG4(S228P), Kappa |
| Application | / |
| Target | Detects C5a |
| Gene | C5a |
| Other Names | C5a, Complement Component 5a |
| Gene ID | 727 (Human) |
| Background | Vilobelimab is a monoclonal antibody that targets C5A, as a first-in-class recombinant chimeric IgG4 antibody, the central mechanism of which is through specific binding and blocking of the biological activity of complement factor C5A, which is the first-in-class recombinant chimeric IgG4 antibody, inhibition of neutrophil chemotaxis, degranulation, and burst of oxygen respiration, thereby controlling the inflammatory response while preserving the innate immune defense function of the membrane attack complex (C5b-9) , to avoid potential safety risks of conventional C5 inhibitors. As a pioneering drug targeting C5A, Vilobelimab has also shown therapeutic potential in neutrophil-mediated inflammatory diseases such as sepsis, acute respiratory distress syndrome (Ards) , ischemia-reperfusion injury, etc. , many countries in the world have launched phase III or Phase II clinical trials, and companies such as China are also deploying similar drug research and development, marking a breakthrough in the field of complement-targeted therapy in immunity and inflammation. |
| Storage | Store at 2-8℃ short term (1-2 weeks).Store at ≤ -20℃ long term. Avoid repeated freeze-thaw. |
| Formulation | Supplied as a 0.2 μm filtered solution of PBS, pH7.2-7.4. |
| Endotoxin | < 1 EU/mg, determined by LAL gel clotting assay |
| Species Reactivity | Human |
| Clone | Vilobelimab |
| Source/Isotype | Human IgG4(S228P), Kappa |
| Application | / |
| Target | Detects C5a |
| Gene | C5a |
| Other Names | C5a, Complement Component 5a |
| Gene ID | 727 (Human) |
| Background | Vilobelimab is a monoclonal antibody that targets C5A, as a first-in-class recombinant chimeric IgG4 antibody, the central mechanism of which is through specific binding and blocking of the biological activity of complement factor C5A, which is the first-in-class recombinant chimeric IgG4 antibody, inhibition of neutrophil chemotaxis, degranulation, and burst of oxygen respiration, thereby controlling the inflammatory response while preserving the innate immune defense function of the membrane attack complex (C5b-9) , to avoid potential safety risks of conventional C5 inhibitors. As a pioneering drug targeting C5A, Vilobelimab has also shown therapeutic potential in neutrophil-mediated inflammatory diseases such as sepsis, acute respiratory distress syndrome (Ards) , ischemia-reperfusion injury, etc. , many countries in the world have launched phase III or Phase II clinical trials, and companies such as China are also deploying similar drug research and development, marking a breakthrough in the field of complement-targeted therapy in immunity and inflammation. |
| Storage | Store at 2-8℃ short term (1-2 weeks).Store at ≤ -20℃ long term. Avoid repeated freeze-thaw. |
| Formulation | Supplied as a 0.2 μm filtered solution of PBS, pH7.2-7.4. |
| Endotoxin | < 1 EU/mg, determined by LAL gel clotting assay |