| Expression System | CHO |
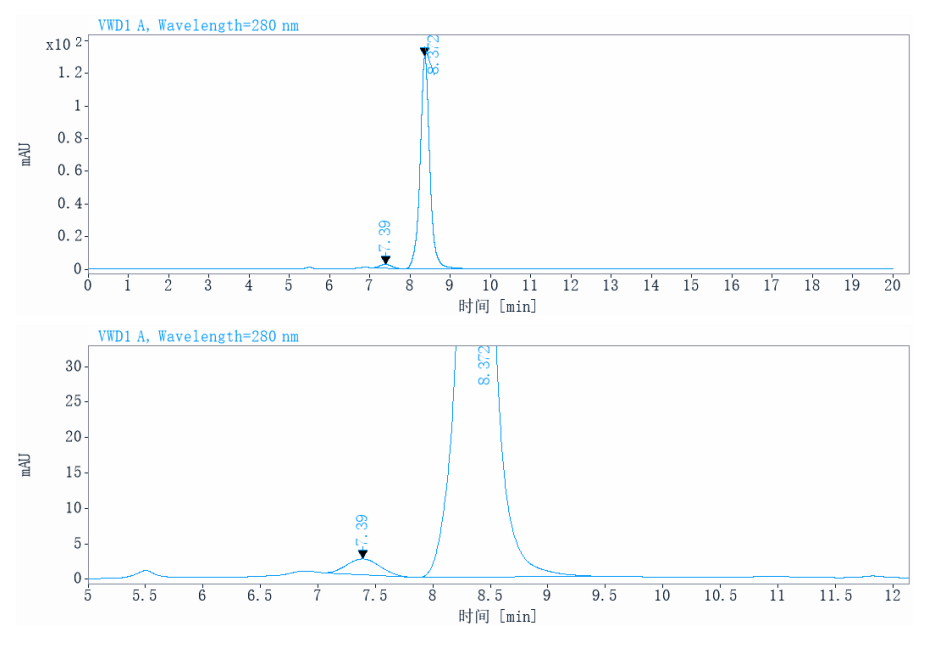
| Aggregation | < 5% as determined by SEC-HPLC |
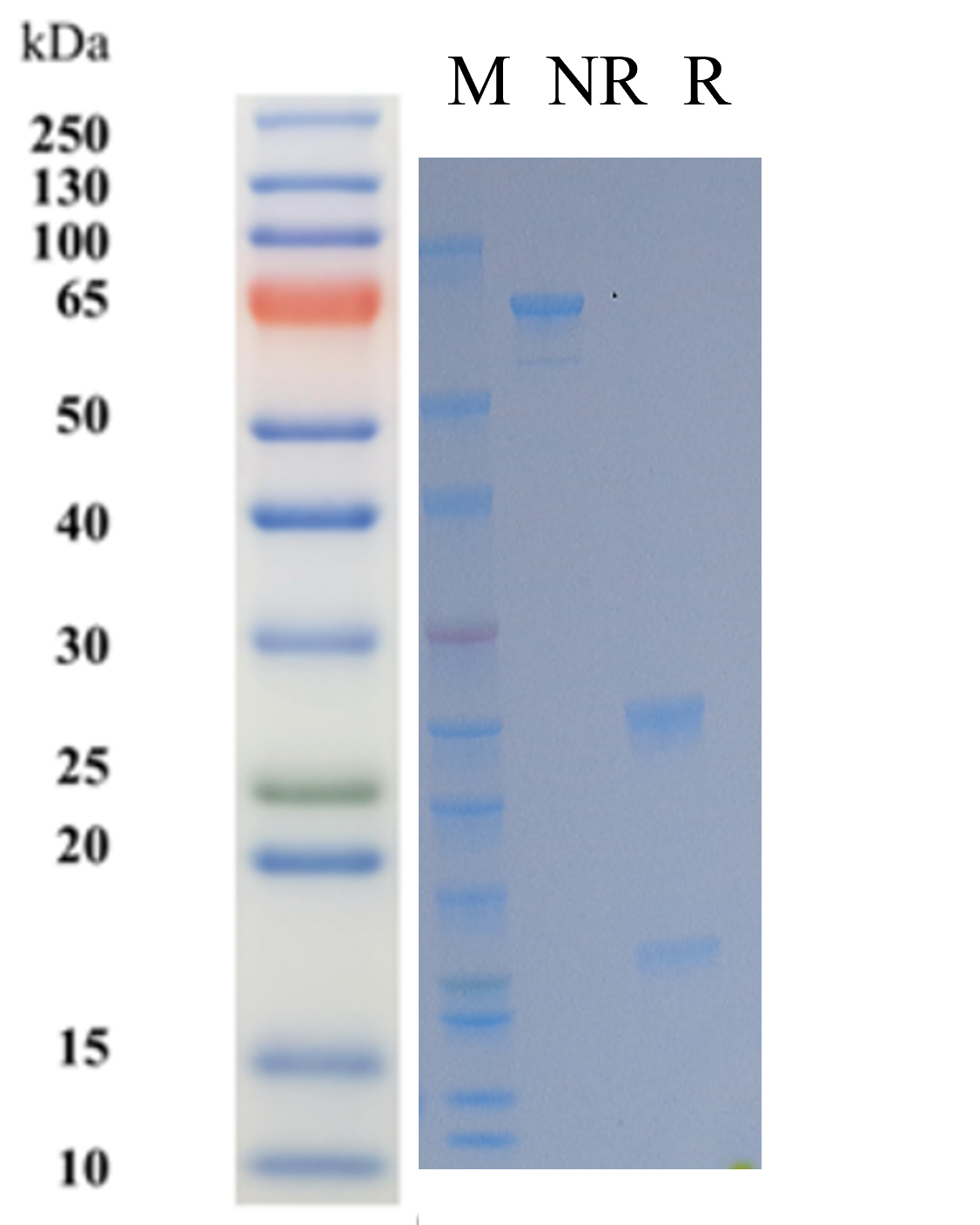
| Purity | > 95% as determined by SDS-PAGE |
| Endotoxin | < 1 EU/mg, determined by LAL gel clotting assay |
| Sterility | 0.2 μm Filtered |
| Target | EGFR&Met |
| Clone | Amivantamab |
| Alternative Names | EGFR: ERBB,ERBB1,HER1,PIG61,mENA Met: MET,AUTS9,HGFR,RCCP2,c-Met |
| Source/Isotype | Human IgG1 (REEM) F405L, K409R ,Kappa |
| Application | Bioactivity-ELISA |
| Description | Amivantamab, an EGFR-MET bispecific monoclonal antibody, is primarily used in the treatment of non-small-cell lung carcinoma (NSCLC) , especially in patients with EGFR Exon 20 insertion mutations (EGFR Exon 20ins) , and as a treatment option after osimertinib resistance. It inhibits tumor cell proliferation and recruits immune cells, such as natural killer cells and macrophages, for anticancer activities by simultaneously blocking EGFR and MET signaling pathways. In clinical trials, Amivantamab in combination with chemotherapy has significantly reduced the risk of tumor progression or death in patients, improved tumor control time and objective response rates, and shown efficacy against brain metastases. In addition, Amivantamab combined with Lazertinib has also shown superior efficacy to osimertinib in the first-line treatment, providing patients with new treatment options. |
| Formulation | phosphate-buffered solution, pH 7.2-7.4. |
| Expression System | CHO |
| Aggregation | < 5% as determined by SEC-HPLC |
| Purity | > 95% as determined by SDS-PAGE |
| Endotoxin | < 1 EU/mg, determined by LAL gel clotting assay |
| Sterility | 0.2 μm Filtered |
| Target | EGFR&Met |
| Clone | Amivantamab |
| Alternative Names | EGFR: ERBB,ERBB1,HER1,PIG61,mENA Met: MET,AUTS9,HGFR,RCCP2,c-Met |
| Source/Isotype | Human IgG1 (REEM) F405L, K409R ,Kappa |
| Application | Bioactivity-ELISA |
| Description | Amivantamab, an EGFR-MET bispecific monoclonal antibody, is primarily used in the treatment of non-small-cell lung carcinoma (NSCLC) , especially in patients with EGFR Exon 20 insertion mutations (EGFR Exon 20ins) , and as a treatment option after osimertinib resistance. It inhibits tumor cell proliferation and recruits immune cells, such as natural killer cells and macrophages, for anticancer activities by simultaneously blocking EGFR and MET signaling pathways. In clinical trials, Amivantamab in combination with chemotherapy has significantly reduced the risk of tumor progression or death in patients, improved tumor control time and objective response rates, and shown efficacy against brain metastases. In addition, Amivantamab combined with Lazertinib has also shown superior efficacy to osimertinib in the first-line treatment, providing patients with new treatment options. |
| Formulation | phosphate-buffered solution, pH 7.2-7.4. |