| Alternative Names | TNFRSF1B, CD120b, TBPII, TNF-R-II, TNF-R75, TNFBR, TNFR1B, TNFR2 |
| Source | Cynomolgus TNFR2 Protein; His Tag (GM-88493RP) is expressed from human 293 cells (HEK-293). It contains AA Leu 23 - Asp 257 (Accession # XP_005544817.1). This protein carries a His tag at the C-terminus. |
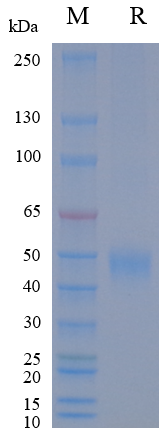
| Purity | > 95% as determined by SDS-PAGE |
| Endotoxin | < 1 EU/μg, determined by LAL gel clotting assay |
| Predicted Mol Mass | 25.9 KDa |
| Formulation | Supplied as a 0.2 μm filtered solution of PBS, pH7.2-7.4. |
| Description | TNF receptors 1 and 2 (TNFR1 and TNFR2) mediate the effects of tumor necrosis factor (TNF). TNFR1, found on most nucleated cells, has a death domain and can trigger apoptosis or NF-κB/MAPK–mediated survival signals, activated by both soluble and membrane-bound TNF. TNFR2, mainly on immune and endothelial cells (lacks a death domain), promotes cell survival and regulatory T cell function via NF-κB and related pathways, often in response to membrane-bound TNF. The two receptors can form heterotrimers and modulate each other’s signaling, reflecting TNF biology’s complexity. TNF-α signals primarily through its receptors TNFR1 (p55) and TNFR2 (p75), triggering multiple pathways. TNFR1 activation commonly leads to NF-κB and MAPK signaling, promoting transcription of inflammatory genes, survival factors, and sometimes apoptosis via caspase cascades. TNFR2 mainly modulates immune cell activation and can enhance TNF-α–driven responses. Cross-talk with other cytokines shapes the intensity and duration of inflammation. Therapeutically, TNF-α inhibitors are used to treat autoimmune diseases, but neutralization must balance infection risk and immune suppression. |
| Alternative Names | TNFRSF1B, CD120b, TBPII, TNF-R-II, TNF-R75, TNFBR, TNFR1B, TNFR2 |
| Source | Cynomolgus TNFR2 Protein; His Tag (GM-88493RP) is expressed from human 293 cells (HEK-293). It contains AA Leu 23 - Asp 257 (Accession # XP_005544817.1). This protein carries a His tag at the C-terminus. |
| Purity | > 95% as determined by SDS-PAGE |
| Endotoxin | < 1 EU/μg, determined by LAL gel clotting assay |
| Predicted Mol Mass | 25.9 KDa |
| Formulation | Supplied as a 0.2 μm filtered solution of PBS, pH7.2-7.4. |
| Description | TNF receptors 1 and 2 (TNFR1 and TNFR2) mediate the effects of tumor necrosis factor (TNF). TNFR1, found on most nucleated cells, has a death domain and can trigger apoptosis or NF-κB/MAPK–mediated survival signals, activated by both soluble and membrane-bound TNF. TNFR2, mainly on immune and endothelial cells (lacks a death domain), promotes cell survival and regulatory T cell function via NF-κB and related pathways, often in response to membrane-bound TNF. The two receptors can form heterotrimers and modulate each other’s signaling, reflecting TNF biology’s complexity. TNF-α signals primarily through its receptors TNFR1 (p55) and TNFR2 (p75), triggering multiple pathways. TNFR1 activation commonly leads to NF-κB and MAPK signaling, promoting transcription of inflammatory genes, survival factors, and sometimes apoptosis via caspase cascades. TNFR2 mainly modulates immune cell activation and can enhance TNF-α–driven responses. Cross-talk with other cytokines shapes the intensity and duration of inflammation. Therapeutically, TNF-α inhibitors are used to treat autoimmune diseases, but neutralization must balance infection risk and immune suppression. |