| Species Reactivity | Human; |
| Clone | Pembrolizumab |
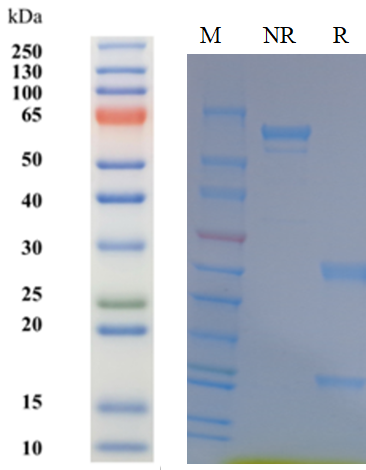
| Source/Isotype | Mouse IgG2b, Kappa |
| Application | / |
| Specificity | Detects PD1 |
| Gene | PD1 |
| Other Names | PDCD1, PD1, CD279, SLEB2 |
| Gene ID | 6622 (human) |
| Background | Early studies with Pembrolizumab, an anti-PD-1 monoclonal antibody, have shown a 40% objective response rate and over 2 years of progression-free survival in patients with melanoma, this data supports that it became the world's first approved anti-PD-1 drug in 2014, initially for unresectable or metastatic melanoma. With the deep understanding of the PD-1/PD-L1 pathway mechanism, the indications of Pembrolizumab have rapidly expanded to more than a dozen solid tumors, such as non-small-cell lung carcinoma, head and neck squamous cell carcinoma, triple-negative breast cancer, etc. , and achieve precision therapy through biomarker screening. Its clinical value is not only reflected in the single-agent efficacy, but also in the combination with chemotherapy, targeted drugs or immune cell therapy, which significantly prolongs the survival of patients. As a benchmark drug in the field of immune checkpoint inhibitors, the development and clinical application of Pembrolizumab have promoted the paradigm shift of cancer treatment from traditional chemotherapy to immunotherapy, it also provides an important reference for the follow-up bispecific monoclonal antibody, ADC drugs and other innovative therapies. |
| Storage | Store at 2-8℃ short term (1-2 weeks).Store at ≤ -20℃ long term. Avoid repeated freeze-thaw. |
| Formulation | Supplied as a 0.2 μm filtered solution of PBS, pH7.2-7.4. |
| Endotoxin | < 1 EU/mg, determined by LAL gel clotting assay |
| Species Reactivity | Human; |
| Clone | Pembrolizumab |
| Source/Isotype | Mouse IgG2b, Kappa |
| Application | / |
| Specificity | Detects PD1 |
| Gene | PD1 |
| Other Names | PDCD1, PD1, CD279, SLEB2 |
| Gene ID | 6622 (human) |
| Background | Early studies with Pembrolizumab, an anti-PD-1 monoclonal antibody, have shown a 40% objective response rate and over 2 years of progression-free survival in patients with melanoma, this data supports that it became the world's first approved anti-PD-1 drug in 2014, initially for unresectable or metastatic melanoma. With the deep understanding of the PD-1/PD-L1 pathway mechanism, the indications of Pembrolizumab have rapidly expanded to more than a dozen solid tumors, such as non-small-cell lung carcinoma, head and neck squamous cell carcinoma, triple-negative breast cancer, etc. , and achieve precision therapy through biomarker screening. Its clinical value is not only reflected in the single-agent efficacy, but also in the combination with chemotherapy, targeted drugs or immune cell therapy, which significantly prolongs the survival of patients. As a benchmark drug in the field of immune checkpoint inhibitors, the development and clinical application of Pembrolizumab have promoted the paradigm shift of cancer treatment from traditional chemotherapy to immunotherapy, it also provides an important reference for the follow-up bispecific monoclonal antibody, ADC drugs and other innovative therapies. |
| Storage | Store at 2-8℃ short term (1-2 weeks).Store at ≤ -20℃ long term. Avoid repeated freeze-thaw. |
| Formulation | Supplied as a 0.2 μm filtered solution of PBS, pH7.2-7.4. |
| Endotoxin | < 1 EU/mg, determined by LAL gel clotting assay |